Enhancement of mixing in a viscous, non-volatile droplet using a contact-free vapor-mediated interaction†
Abstract
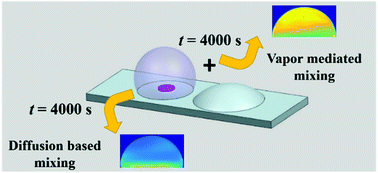
Mixing at small fluidic length scales is especially challenging in viscous and non-volatile droplets frequently encountered in bio-chemical assays. In situ methods of mixing, which depend on diffusion or evaporation-driven capillary flow, are typically slow and inefficient, while thermal or electro-capillary methods that are either complicated to implement or may cause sample denaturing. This article demonstrates an enhanced mixing timescale in a sessile droplet of glycerol by simply introducing a droplet of ethanol in its near vicinity. The fast evaporation of ethanol introduces molecules in the proximity of the glycerol droplet, which are preferentially adsorbed (more on the side closer to ethanol) creating a gradient of surface tension driving the Marangoni convection in the droplet. We conclusively show that for the given volume of the droplet, the mixing time reduces by ∼10 hours due to the vapour-mediated Marangoni convection. Simple scaling arguments are used to predict the enhancement of the mixing timescale. Experimental evidence obtained from fluorescence imaging is used to quantify mixing and validate the analytical results. This is the first proof of concept of enhanced mixing in a viscous, sessile droplet using the vapour mediation technique.


 Please wait while we load your content...
Please wait while we load your content...