The effect of weighted averages when determining the speciation and structure–property relationships of europium(iii) dipicolinate complexes†
Abstract
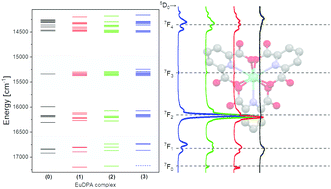
Lanthanide(III) coordination chemistry in solution is inherently complicated by the lack of directional interactions and rapid ligand exchange. The latter can be eliminated in kinetically inert complexes, but remains a challenge in complexes between lanthanide(III) ions and smaller ligands. As multiple conformations and partial decomplexation is an issue even with multidentate ligands, it will influence the observed solution properties of complexes of smaller ligands common in the field of f-elements coordination chemistry such as acetylacetonates and dipicolinates. Here, europium(III) complexes with one, two and three dipicolinates were investigated in a series of 13 samples, where the composition was varied from 0 to 3 equivalents of dipicolinate. While the results did show the formation of three distinct europium(III) dipicolinate complexes confirming the literature data on the system, clear discrepancies in speciation related properties were evident when comparing the results from absorption and luminescence spectroscopy. It was concluded that the difference is due to the difference in time constant of the two experiments. Furthermore, it is shown that the information obtained from luminescence arises from a weigthed average, and with discepancies between the observed and actual concentration exceeding 25%, it is advised that the weighted averages are taken into consideration when reporting on solution properties of lanthanide(III) complexes. From the resolved optical spectra of [Eu(H2O)9]3+, [Eu(DPA)(H2O)6]+, [Eu(DPA)2(H2O)3]−, and [Eu(DPA)3]3−, the excited energy levels and transition probabilities are determined, and it was concluded that both transition probabilities and ligand field effects on the microstates are different in all four species.


 Please wait while we load your content...
Please wait while we load your content...