Elucidating tuneable ambipolar charge transport and field induced bleaching at the CH3NH3PbI3/electrolyte interface†
Abstract
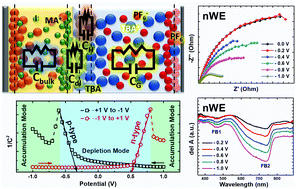
Charge transport through lead trihalide based perovskites is more complex than through any other semiconducting materials due to their mixed electronic–ionic conductivity as well as ambipolarity. Here we have investigated charge transport through a perovskite/electrolyte interface using electrochemical impedance spectroscopy (EIS) under illumination and applied bias conditions. Similar trends in EIS were observed with positively biased as well as negatively biased working electrodes, indicating ambipolar charge transport through the perovskite layer. Ionic conductivity upon photo-illumination plays a significant role in modulating charge transport at the interface by creating an additional built-in field which flips its polarity at a moderate applied bias voltage. Therefore, anomalous charge transport resistance is observed under illumination at around 400 to 600 mV applied bias. Electric field induced UV-Vis absorption spectroscopy shows a decrease in absorption when both positive and negative bias voltages are applied to a perovskite coated ITO working electrode, indicating the occurrence of excited state charge transfer at the electrolyte interface. Two distinct electric field induced bleaching bands have been observed at around 480 nm and 750 nm, similar to the two photobleaching bands due to the dual nature of the excited states in lead halide perovskites.


 Please wait while we load your content...
Please wait while we load your content...