Clarifying the impacts of surface hydroxyls on CO oxidation on CeO2(100) surfaces: a DFT+U study†
Abstract
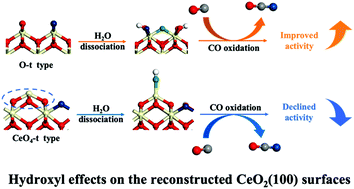
In heterogeneous catalysis, surface hydroxylation is well recognized as a common phenomenon under realistic reaction conditions. However, even for the versatile ceria-based materials that have attracted extensive studies, the results and causes of the hydroxyl effect on the catalytic reactivity remain largely elusive. In this work, density functional theory calculations corrected by on-site Coulomb interaction were conducted to clarify the CO oxidation pathways and also the impacts of surface hydroxyls on the catalytic performance at the two most stable reconstructions of CeO2(100). It is found that the presence of hydroxyl groups can boost the CO oxidation activity on the O-terminal surface but shows an opposite effect on the CeO4-terminal one. Further analyses regarding the structural distortions, electronic structures and orbital interactions reveal that the stretched Ce–O distance via in-plane hydrogen bonds and the electron redistributions induced by additional hydroxyl coordination are the main reasons for the different hydroxyl effects on the O- and CeO4-terminal surfaces. Our results not only uncover the dual-character of surface hydroxyls in heterogeneous catalysis, but they also underline the significance of moderate moisture in the reaction system that may endow ceria catalysts with both good thermostability and high catalytic activity.
- This article is part of the themed collection: 2020 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...