On the wetting translucency of hexagonal boron nitride†
Abstract
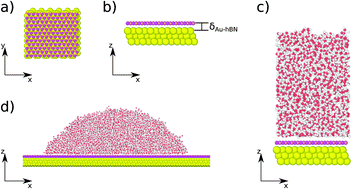
When a liquid drop sits on an atomically thin layer of a 2D van der Waals (vdW) solid (like graphene) supported by a hydrophilic material, it is possible that the drop demonstrates an equilibrium contact angle that is influenced by this underlying hydrophilic material and hence is different from that observed on the bulk 2D material (e.g., graphite) surface. Such a behavior is known as the wetting translucency effect. While the wetting translucency effect of graphene has been extensively studied, the wetting translucency of hexagonal boron nitride (hBN) remains largely unexplored despite significant similarities in structural properties between these materials. In this study, we probe the wetting translucency of hBN. For this purpose, we conduct molecular dynamics simulations of water droplets and water films on hBN layers supported on a gold-like hydrophilic substrate. Our results show that for a substrate coated by monolayer hBN (“coated substrate”), depending on the contact distance between underlying substrate and hBN, an increase in the hydrophilicity of the underlying surface causes a monotonic increase in the overall adhesion energy between water and the coated substrate and a monotonic decrease in the contact angle of a drop on the coated substrate. For an increasing number of stacked hBN layers, the wettability of coated substrate becomes independent of the wettability of the underlying solid. Accordingly, our results confirm a distinct wetting translucency nature of hBN very similar to that observed in graphene.


 Please wait while we load your content...
Please wait while we load your content...