Molecular dynamics simulations of alkaline earth metal ions binding to DNA reveal ion size and hydration effects†
Abstract
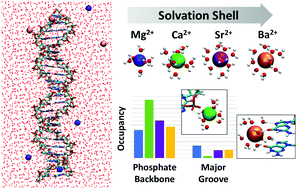
The identity of metal ions surrounding DNA is key to its biological function and materials applications. In this work, we compare atomistic molecular dynamics simulations of double strand DNA (dsDNA) with four alkaline earth metal ions (Mg2+, Ca2+, Sr2+, and Ba2+) to elucidate the physical interactions that govern DNA–ion binding. Simulations accurately model the ion–phosphate distance of Mg2+ and reproduce ion counting experiments for Ca2+, Sr2+, and Ba2+. Our analysis shows that alkaline earth metal ions prefer to bind at the phosphate backbone compared to the major groove and negligible binding occurs in the minor groove. Larger alkaline earth metal ions with variable first solvation shells (Ca2+, Sr2+, and Ba2+) show both direct and indirect binding, where indirect binding increases with ion size. Mg2+ does not fit this trend because the strength of its first solvation shell predicts indirect binding only. Ions bound to the phosphate backbone form fewer contacts per ion compared to the major groove. Within the major groove, metal ions preferentially bind to guanine–cystosine base pairs and form simultaneous contacts with the N7 and O6 atoms of guanine. Overall, we find that the interplay among ion size, DNA–ion interaction, and the size and flexibility of the first solvation shell are key to predicting how alkaline earth metal ions interact with DNA.


 Please wait while we load your content...
Please wait while we load your content...