Tuning metal oxide defect chemistry by thermochemical quenching†
Abstract
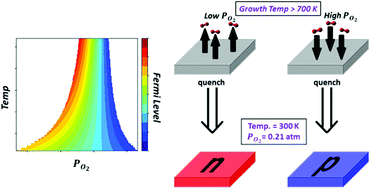
The low-temperature defect chemistry of monoclinic and tetragonal ZrO2 and hematite Fe2O3 is studied in the non-equilibrium state of thermochemical quenching; that is, rapid cooling starting from a certain high temperature and oxygen chemical potential. This non-equilibrium state is of great interest because many metal oxides are used at low temperatures below their growth temperatures. This paper addresses the importance of considering this non-equilibrium state rather than applying equilibrium thermodynamics as commonly used when studying point defects from first principles. Based on point defect formation energies calculated previously using density functional theory, we compare the type of dominant defects at equilibrium to those at a quenched state originating from a certain initial growth temperature and oxygen partial pressure. The comparison is facilitated by casting the dominant defects in a dominance diagram on the temperature – oxygen partial pressure plane. We consider two scenarios to model the quenched state. In the first, only electronic defects equilibrate whereas all ionic defects are frozen. Whereas, in the second, electronic defects and interstitials are allowed to equilibrate under the assumption of mobile interstitials at low temperatures. We find that new ionic charge compensation modes can appear on the dominance diagram after quenching. Additionally, purely ionic charge compensation modes consisting of vacancies and/or interstitials expand in the dominance diagram at the expense of purely electronic compensation modes. For the ZrO2 phases, we argue that scenario 2 is more realistic and leads to difficulty in achieving n-type doping by thermochemical quenching. For Fe2O3, and regardless of the quenching scenario, iron vacancies occupy a wider zone of domination, which limits the performance of this oxide as a water splitting photoanode. Our study shows that by controlling the growth thermochemical conditions, it is possible to tune the Fermi level of oxides over a considerable range within the band gap by quenching. This provides an extra tool to tune the electric conductivity of metal oxides beyond traditional extrinsic doping. This work indicates that non-equilibrium thermodynamic analysis is necessary to understand and control defect chemistry at low temperatures.

 Please wait while we load your content...
Please wait while we load your content...