A DFT study on a borophene/boron nitride interface for its application as an electrode
Abstract
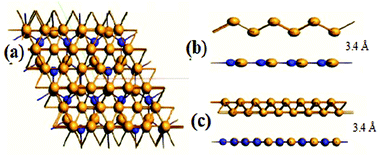
In order to search for a new anode material for lithium-ion batteries (LIBs), a borophene/boron nitride (B/BN) interface was investigated in detail using density functional theory. Borophene is an excellent two-dimensional (2D) anode material that offers high charging capacity and a low energy barrier, but it suffers from stability issues when it is used in its free-standing form. The findings of this work indicate that the thermal and mechanical stabilities of the borophene epilayer are notably increased by preparing its interface with a boron nitride substrate. The electronic properties of the lithiated and delithiated interface exhibited metallic behavior, whereas the mechanical stiffness of the interface increased three times when compared with that of the pristine borophene. The thermal stability was calculated by molecular dynamics and indicated a six times increase in its value for the interface. The interface exhibited a specific charging capacity of 1698 mA h g−1, which is higher than that of bare borophene and several other 2D materials. Furthermore, nudged elastic band (NEB) calculations indicated a low energy barrier to diffusion of Li in the interface. These advantages of the B/BN interface make it an excellent choice as an anode material for LIBs.


 Please wait while we load your content...
Please wait while we load your content...