Li-ion transport at the interface between a graphite anode and Li2CO3 solid electrolyte interphase: ab initio molecular dynamics study†
Abstract
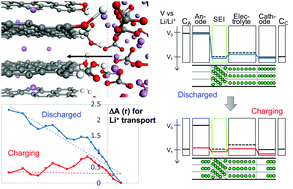
Understanding and the control of Li-ion (Li+) transport across the interface between the anode and solid electrolyte interphase (SEI) film or electrolyte is a key issue in battery electrochemistry and interface science. In this study, we investigated the structural, electronic and free energy properties of Li+ migration between a Li-intercalated graphite anode LiCx and Li2CO3 SEI film, by using ab initio molecular dynamics and free energy calculations. We compared three types of graphite edges: H-, OH- and mixed (H, OH, COOH)-terminations, and three cases of transferred Li-ions: Li+ constructing the SEI, excess Li+ and excess Li0 (excess Li+ + e− in anode). After validation of our calculations with Li2CO3 and LiCx bulk systems, we sampled the interfacial structures under thermodynamic equilibrium and demonstrated that the OH- and mixed-terminations had larger binding energies. The calculated free energy profiles of Li+ intercalation from the Li2CO3 SEI to LiC24 showed barriers larger than 1.2 eV irrespective of the terminations and Li+ cases. We also clarified that the charges of Li ions did not change much upon the intercalation. Based on these results and the calculated Li chemical potential, we constructed the probable free energy profile of Li+ between the anode and cathode under charging and discharging. This profile model suggest a possible electric field approximation for the charging stage, and the resultant free energy profiles with such fields gave a ca. 0.5 eV barrier under charging, which was consistent with the experimental values. The present picture will give a crucial insight into Li-ion transport at the battery interfaces.
- This article is part of the themed collection: Frontiers in Molecular Simulation of Solvated Ions, Molecules and Interfaces


 Please wait while we load your content...
Please wait while we load your content...