Orpiment under compression: metavalent bonding at high pressure†
Abstract
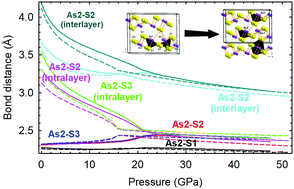
We report a joint experimental and theoretical study of the structural, vibrational, and electronic properties of layered monoclinic arsenic sulfide crystals (α-As2S3), aka mineral orpiment, under compression. X-ray diffraction and Raman scattering measurements performed on orpiment samples at high pressure and combined with ab initio calculations have allowed us to determine the equation of state and the tentative assignment of the symmetry of many Raman-active modes of orpiment. From our results, we conclude that no first-order phase transition occurs up to 25 GPa at room temperature; however, compression leads to an isostructural phase transition above 20 GPa. In fact, the As coordination increases from threefold at room pressure to more than fivefold above 20 GPa. This increase in coordination can be understood as the transformation from a solid with covalent bonding to a solid with metavalent bonding at high pressure, which results in a progressive decrease of the electronic and optical bandgap, an increase of the dielectric tensor components and Born effective charges, and a considerable softening of many high-frequency optical modes with increasing pressure. Moreover, we propose that the formation of metavalent bonding at high pressures may also explain the behavior of other group-15 sesquichalcogenides under compression. In fact, our results suggest that group-15 sesquichalcogenides either show metavalent bonding at room pressure or undergo a transition from p-type covalent bonding at room pressure towards metavalent bonding at high pressure, as a precursor towards metallic bonding at very high pressure.


 Please wait while we load your content...
Please wait while we load your content...