The corrosion inhibition of stainless steel by ferrocene–polyoxometalate hybrid molecular materials – experimental and first principles studies†
Abstract
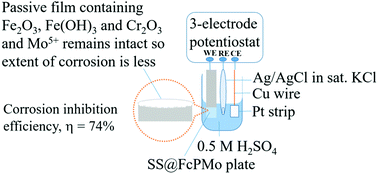
The hybrid molecular materials [Fe(C5H5)2]3[PW12O40]·(H2O)4 and [Fe(C5H5)2]3[PMo12O40]·(H2O)6, denoted respectively as FcPW and FcPMo, are synthesized via a co-precipitation method under acidic conditions (pH: ∼2–3). The molecular properties of these compounds are investigated via various analytical methods. UV-vis diffuse reflectance and photoluminescence studies confirm that these compounds are charge-transfer salts. Stainless steel plates (SS, 316 grade) coated with these hybrid compounds and dipped in 0.5 M H2SO4 and Ringer's solutions show significantly inhibited corrosion. The corrosion inhibition properties of these materials are studied via weight loss, electrochemical impedance spectroscopy, and potentiodynamic polarization studies. The reproducibility of the corrosion inhibition results is ensured via repeat measurements, and error values are reported. The surfaces of the corroded steel plates are examined via scanning electron and atomic force microscopy techniques. It is found that the corrosion is of uniform type. The Tafel extrapolation method shows that the inhibition efficiency is 74% for SS@FcPMo and 65% for SS@FcPW. The corrosion inhibition mechanism is explained based on the redox nature of the hybrid materials. The experimental results are corroborated through computational studies carried out using a first principles approach with the optimized geometries and frontier molecular orbitals of the hybrid molecular materials.


 Please wait while we load your content...
Please wait while we load your content...