In situ observation of the potential-dependent structure of an electrolyte/electrode interface by heterodyne-detected vibrational sum frequency generation†
Abstract
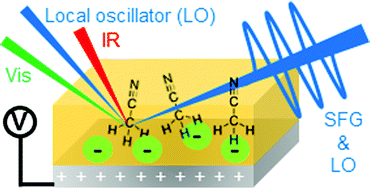
Elucidating the structure of electrolyte/electrode interfaces is of essential importance not only for understanding of the fundamental process of electrochemistry but also for developing next-generation rechargeable batteries. In this study, we applied HD-VSFG spectroscopy to study a prototypical non-aqueous electrochemical interface of a platinum electrode in 0.1 M LiCF3SO3 acetonitrile (CH3CN) solution, and measured Im χ(2) spectra by changing the applied potential in the range of −0.8 V to 2.0 V. In the positive potential region, the positive bands assignable to acetonitrile appear in the CH3 and CN stretch regions, and their positive signs indicate the CH3-down orientation of acetonitrile at the interface. We also observed an SO3− stretch band of the anion of the electrolyte and found that the potential dependence of its intensity is similar to those of the CH3 and CN bands of acetonitrile. These observations indicate that the CF3SO3− anion is adsorbed at the platinum surface in the positive potentials, which induces CH3-down orientation of acetonitrile at the interface. The present study demonstrates the advantages of HD-VSFG spectroscopy for studying electrochemical systems, and it opens a new way to investigate electrolyte/electrode interfaces at the molecular level.


 Please wait while we load your content...
Please wait while we load your content...