Record-high stability and compactness of multiply-charged clusters aided by selected terminal groups†
Abstract
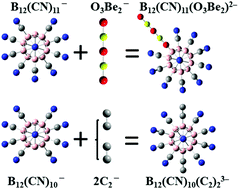
Multiply-charged clusters with compact sizes that are stable in the gas phase are important due to their potential applications as weakly-coordinating ions and building blocks of bulk materials. However, the number of these clusters, especially those with high charge states beyond two, is limited. In this work, we show that gas-phase di- and tri-anions with record-high stability and compactness can be developed by utilizing a series of stable mono-anions with linear configurations as ligands. A stable di-anion with a record-high second electron binding energy of 6.26 eV and the smallest tri-anion with a record-low radius of 4.85 Å have been identified. This study demonstrates that multiply-charged clusters with highly charged state and compact sizes can be stabilized solely by selected terminal groups in the structure. This finding significantly enriches the database of stable multiply-charged gas-phase clusters and provides a path towards rational design of further multiply-charged species.


 Please wait while we load your content...
Please wait while we load your content...