Decomposition mechanism scenarios of CL-20 co-crystals revealed by ReaxFF molecular dynamics: similarities and differences†
Abstract
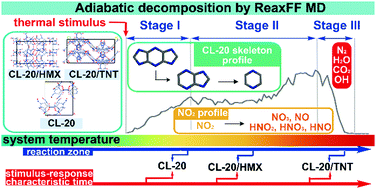
Understanding the similarities and differences of decomposition mechanisms of CL-20 and its cocrystals is of great interest for practical applications of CL-20 cocrystals. The responses of CL-20 cocrystals to thermal stimulus were investigated using ReaxFF molecular dynamics simulations of two representative cocrystals, CL-20/HMX and CL-20/TNT, under adiabatic conditions and comparing to the baseline system of pure CL-20. The comprehensive chemical details were revealed with the aid of the unique code of VARxMD. The three CL-20-involved reactive systems all exhibit a distinct three-stage character during adiabatic decomposition when using the double peaks of the major intermediate NO2 amount as the boundary. By taking advantage of the three-stage classification, a clear scenario for the similar stimulus-response of the CL-20 cocrystals can be elucidated for the dominant primary decomposition of CL-20 in stage I and the transition of favored chemical mechanisms from the generation of intermediates/radicals in stage II into their consumption to form stable products in stage III. The similar chemical behaviors are rooted in the dominance of CL-20 chemistry in the initial response of its cocrystals to thermal stimulus. The prolonged reaction zone uncovers the slowed decomposition kinetics of CL-20/HMX and CL-20/TNT, which is associated with the altered kinetics of CL-20 decomposition specifically by N–NO2 bond scission and CL-20 skeleton decay. The retarded CL-20 decomposition in its cocrystals consequently results in more moderate self-heating and less violent exothermic reactions that agrees with the experimental observations of improved stability and damaged detonation performance of CL-20 cocrystals, particularly for CL-20/TNT. The results obtained in this work suggest that ReaxFF MD simulations can provide useful insight for the modulated chemical properties of varied CL-20 cocrystals.
- This article is part of the themed collection: 2020 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...