The synergistic role of the support surface and Au–Cu alloys in a plasmonic Au–Cu@LDH photocatalyst for the oxidative esterification of benzyl alcohol with methanol†
Abstract
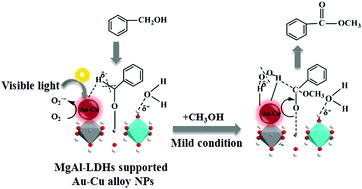
Layered double hydroxide-supported Au–Cu alloy nanoparticles (NPs) were found to be highly efficient catalysts for the oxidative esterification of benzyl alcohol with methanol in the presence of molecular oxygen under visible-light irradiation to prepare methyl benzoate. Here, we report that alloying small amounts of copper into gold nanoparticles can increase the ability to activate oxygen molecules to O2˙− radicals and display greater charge heterogeneity to promote the cleavage of the C–H bond of benzyl alcohol molecules by reinforcing the coordination of the intermediate with unsaturated metal active sites due to the LSPR effect of alloy NPs, which is the rate-limiting step of the reaction. Besides the Au–Cu alloy NPs, the support also played a pivotal role in the catalytic process. The support with the presence of acid–base pairs, in which the basic sites served as the reactant molecule adsorption sites to provoke the intermediate formation and the acidic sites promoted the recovery of the support surface, showed better performances by affecting the overall reaction rate completely. Moreover, applying this photocatalyst in the cross-esterification of aromatic alcohols and aliphatic alcohols displayed excellent yields.


 Please wait while we load your content...
Please wait while we load your content...