5-Fold symmetry in superatomic scandium clusters: exploiting favourable orbital overlap to sequester spin
Abstract
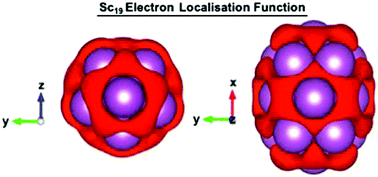
The geometries and electronic structures of icosahedral A13C (A = Sc, Y; C = 0, ±1, ±2) clusters have been determined at a range of multiplicities at each cluster charge, using density functional theory methods. These clusters demonstrate a complex electronic structure which provides insight into the anomalously high magnetic moment of icosahedral group 3 clusters and further contextualises the role of transition metals and d-electrons within the superatomic model. Embedded deeply within the density of states for these clusters are typical superatom orbitals which are populated up to the 2S level. Above the 2S-state there are three states of apparent F symmetry, which are preferentially singly occupied, followed by an abundance of approximately degenerate P-, G-, D- and F-states at the Fermi energy, which are at most singly occupied. In spite of apparent angular symmetry and a nodal structure reminiscent of superatomic orbitals these states are actually formed from preferential overlap of the valence d-orbitals of the cluster atoms. This analysis was further contextualised through analysis of the Sc19 cluster, which shows a similar construction of Kohn–Sham states, but with the breaking of 5-fold symmetry along one of its Cartesian axes. Finally, this work clearly demonstrates the ability of d-electrons to give rise to superatomic orbitals is not just constrained by atomic species but also by the local environment of the atoms.


 Please wait while we load your content...
Please wait while we load your content...