Improved SERS activity of non-stoichiometric copper sulfide nanostructures related to charge-transfer resonance†
Abstract
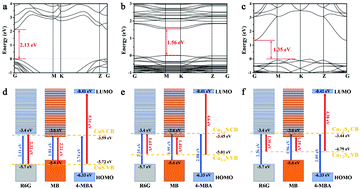
The low enhancement factor of semiconductor SERS substrates is a major obstacle for their practical application. Therefore, there is a need to explore the facile synthesis of new SERS substrates and reveal the SERS enhancement mechanism. Here, we develop a simple, facile and low-cost two-step method to synthesize copper sulfide based nanostructures with different Cu7.2S4 contents. The as-synthesized sample is composed of nanosheets with the CuS phase structure. With the increase of the annealing temperature to 300 °C, the CuS content gradually decreases and disappears, and the content of Cu7.2S4 and CuSO4 appears and gradually increases. At the annealing temperature of 350 °C, only CuSO4 exists. Compared with pure CuS or pure CuSO4, the detection limit of R6G molecules is the lowest for the composite sample with a higher content of Cu7.2S4, indicating that the introduction of non-stoichiometric Cu7.2S4 can improve the SERS performance and the higher content of Cu7.2S4 leads to a higher SERS activity. Furthermore, to investigate the SERS mechanism, the energy band structures and energy-level diagrams of different probe molecules over CuS, Cu7.2S4 and CuxS are studied by DFT calculations. Theoretical calculations indicate that the excellent SERS behavior depends on charge transfer resonance. Our work provides a general approach for the construction of excellent metal compound semiconductor SERS active substrates.


 Please wait while we load your content...
Please wait while we load your content...