Ion–ion interactions enhance aluminum solubility in alkaline suspensions of nano-gibbsite (α-Al(OH)3) with sodium nitrite/nitrate†
Abstract
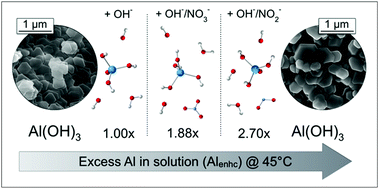
Despite widespread industrial importance, predicting metal solubilities in highly concentrated, multicomponent aqueous solutions is difficult due to poorly understood ion–ion and ion–solvent interactions. Aluminum hydroxide solid phase solubility in concentrated sodium hydroxide (NaOH) solutions is one such case, with major implications for ore refining, as well as processing of radioactive waste stored at U.S. Department of Energy legacy sites, such as the Hanford Site, Washington State. The solubility of gibbsite (α-Al(OH)3) is often not well predicted because other ions affect the activity of hydroxide (OH−) and aluminate (Al(OH)4−) anions. In the present study, we systematically examined the influence of key anions, nitrite (NO2−) and nitrate (NO3−), as sodium salts on the solubility of α-Al(OH)3 in NaOH solutions taking care to establish equilibrium from both under- and oversaturation. Rapid equilibration was enabled by use of a highly pure and crystalline synthetic nano-gibbsite of well-defined particle size and shape. Measured dissolved aluminum concentrations were compared with those predicted by an α-Al(OH)3 solubility model derived for simple Al(OH)4−/OH− systems. Specific anion effects were expressed as an enhancement factor (Alenhc) conveying the excess of dissolved aluminum. At 45 °C, NaNO2 and NaNO3-containing systems exhibited Alenhc values of 2.70 and 1.88, respectively, indicating significant enhancement. The solutions were examined by Raman and high-field 27Al NMR spectroscopy, indicating specific interactions including Al(OH)4−–Na+ contact ion pairing and Al(OH)4−–NO2−/NO3− ion–ion interactions. Dynamic evolution of the α-Al(OH)3 particles including growth and agglomeration was observed revealing the importance of dissolution/reprecipitation in establishing equilibrium. These studies indicate that incomplete ion hydration, as a result of the low water activity in these concentrated electrolytes, results in: (i) enhanced reactivity of the hydroxide ion with respect to α-Al(OH)3; (ii) increased concentrations of Al(OH)4− in solution; and (iii) stronger ion–ion interactions that act to stabilize the supersaturated solutions. This information on the mechanisms by which α-Al(OH)3 becomes supersaturated is essential for more energy-efficient aluminum processing technologies, including the treatment of millions of gallons of Al(OH)4−-rich high-level radioactive waste.
- This article is part of the themed collection: 2019 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...