Investigation of the mechanism of overall water splitting in UV-visible and infrared regions with SnC/arsenene vdW heterostructures in different configurations†
Abstract
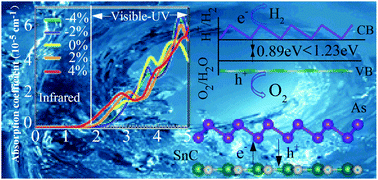
Monolayer arsenene presents good stability and high photogenic carrier mobility. However, a high photogenic electron and hole pair recombination rate seriously reduces its photocatalytic activity. The photocatalytic activity can be effectively improved by building type II heterostructures. In this work, SnC/arsenene heterostructures in three configurations are studied using first-principles calculations. Their structure, stability, and electronic and photocatalytic properties are investigated. It is found that all SnC/arsenene heterostructures are stable, and form type-II band edges, which effectively promote the transfer of photogenerated electrons from the SnC monolayer to the arsenene sheet. The charge transfer between SnC and arsenene leads to a built-in electric field in the interface region, which is favorable for inhibiting photogenic electron and hole pair recombination. Compared with the monolayer arsenene, the photocatalytic activity is greatly improved and the absorption spectrum of SnC/arsenene heterostructures is expanded. Attractively, these three heterostructures present two different photocatalytic mechanisms. H1 and H3 configurations were taken as examples to study their photocatalytic properties for overall water splitting at varying pH values and external strains. We found that alkaline conditions were more favorable for photocatalysis of SnC/arsenene heterostructures. In particular, H3 can still achieve full photocatalytic water decomposition in the near infrared region. These results show that the SnC/arsenene heterostructures are a prospective material for photocatalysis application.
- This article is part of the themed collection: 2019 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...