A guinea pig for conformer selectivity and mechanistic insights into dissociative ionization by photoelectron photoion coincidence: fluorocyclohexane
Abstract
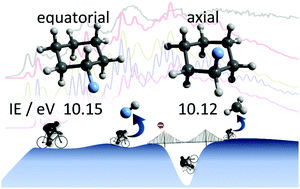
We studied fluorocyclohexane (C6H11F, FC6) by double imaging photoelectron photoion coincidence spectroscopy in the 9.90–13.90 eV photon energy range. The photoelectron spectrum can identify species isomer and, in this case, even conformer selectively. Ab initio results indicated that the axial conformer has two, close-lying cation electronic states. With the help of Franck–Condon simulations of the vibrational fine structure, we determined the origin of three transitions, (i) axial FC6 → axial FC6+ of C1 symmetry (![[X with combining tilde]](https://www.rsc.org/images/entities/b_char_0058_0303.gif) +, A′′ in CS), (ii) equatorial FC6 → equatorial FC6+ of C1 symmetry (
+, A′′ in CS), (ii) equatorial FC6 → equatorial FC6+ of C1 symmetry (![[X with combining tilde]](https://www.rsc.org/images/entities/b_char_0058_0303.gif) +, A′′ in CS), and (iii) axial FC6 → A′ axial FC6+ of CS symmetry (Ã+) as 10.12 ± 0.01, 10.15 ± 0.01 and 10.15 ± 0.02 eV, respectively. At slightly higher energies, the FC6 cation starts fragmenting by HF loss (E0 = 10.60 eV), followed by sequential CH3 (E0 = 10.71 eV) or C2H4 (E0 = 11.06 eV) loss. Surprisingly, the methyl-loss step has an effective barrier of only 0.11 eV, and yet it is a slow process at threshold. Based on the statistical model, this is explained by isomerization and stabilization of the C6H10+ intermediate. The highest energy channel observed, vinyl fluoride (C2H3F) loss yielding C4H8+ appears in the breakdown diagram at 12 eV, which agrees with the computed threshold to cyclobutane cation formation. However, the model predicted a ca. 1 eV competitive shift for this parallel channel, i.e., an E0 = 11.23 eV. This led us to explore the potential energy surface to find a lower-lying fragmentation channel including H-transfer steps. Rate constant measurements and statistical modeling thus yield fundamental insights into the reaction mechanism beyond what is immediately seen in the mass spectra.
+, A′′ in CS), and (iii) axial FC6 → A′ axial FC6+ of CS symmetry (Ã+) as 10.12 ± 0.01, 10.15 ± 0.01 and 10.15 ± 0.02 eV, respectively. At slightly higher energies, the FC6 cation starts fragmenting by HF loss (E0 = 10.60 eV), followed by sequential CH3 (E0 = 10.71 eV) or C2H4 (E0 = 11.06 eV) loss. Surprisingly, the methyl-loss step has an effective barrier of only 0.11 eV, and yet it is a slow process at threshold. Based on the statistical model, this is explained by isomerization and stabilization of the C6H10+ intermediate. The highest energy channel observed, vinyl fluoride (C2H3F) loss yielding C4H8+ appears in the breakdown diagram at 12 eV, which agrees with the computed threshold to cyclobutane cation formation. However, the model predicted a ca. 1 eV competitive shift for this parallel channel, i.e., an E0 = 11.23 eV. This led us to explore the potential energy surface to find a lower-lying fragmentation channel including H-transfer steps. Rate constant measurements and statistical modeling thus yield fundamental insights into the reaction mechanism beyond what is immediately seen in the mass spectra.


 Please wait while we load your content...
Please wait while we load your content...