Strain effects on Co,N co-decorated graphyne catalysts for overall water splitting electrocatalysis†
Abstract
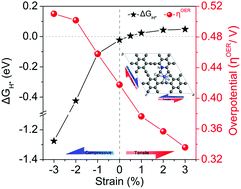
Lattice strain, either tensile or compressive, can fine-tune the electronic structure of surfaces via altering the distances between surface atoms, thereby modifying the catalytic activity of catalysts. Numerous examples of strain engineering have been applied to various electrocatalysts for both hydrogen evolution reaction (HER) and oxygen evolution reaction (OER), but strain-optimized 2D carbon-based single-atom electrocatalysts for catalyzing the overall water splitting reaction have received little attention. Here, we applied the lattice strain of Co,N co-decorated graphyne (Co@N1-GY) to directly optimize its catalytic activity for the overall water splitting reaction based on first-principles calculations. Our calculations suggest that compressive strain and tensile strain lead to less stability of Co@N1-GY and the distances between C and Co atoms increase linearly with the strain changing from compressive to tensile, thus linearly upshifting the p-band center of C atoms and the d-band center of Co atoms. In addition, biaxial strain has more remarkable effects on these properties than uniaxial strain. From compressive to tensile strain, the chemisorption of electrochemically generated intermediates in both HER and OER becomes weaker and weaker. A tensile strain of 0.5% on Co@N1-GY gives an ideal HER performance, while the OER reaches the minimum overpotential of 0.33 V under the biaxial tensile strain of 3%.


 Please wait while we load your content...
Please wait while we load your content...