Microwave plasma-based direct synthesis of free-standing N-graphene
Abstract
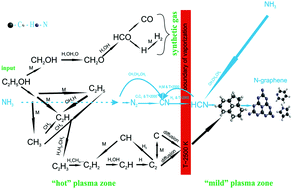
Free-standing N-graphene was synthesized using a microwave plasma-based method at atmospheric pressure conditions through a single step and in a controllable manner. Using ethanol and ammonia as precursors, N-graphene with low relative amount of bonded oxygen and low level of saturated sp3 carbon bonds was produced. Adjusting the injection position of the nitrogen precursor in the plasma medium leads to selectivity in terms of doping level, nitrogen configuration and production yield. A previously developed theoretical model, based on plasma thermodynamics and chemical kinetics, was further updated to account for the presence of nitrogen precursor. The important role of HCN attachment to the graphene sheets as the main process of N-graphene formation is elucidated. The model predictions were validated by experimental results. Optical Emission Spectroscopy was used to detect the emission of plasma generated “building units” and to determine the gas temperature. The plasma outlet gas was analyzed by Fourier-Transform Infrared Spectroscopy to detect the generated gaseous by-products. The synthesized N-graphene was characterized by Scanning Electron Microscopy, Raman and X-ray photoelectron spectroscopies.


 Please wait while we load your content...
Please wait while we load your content...