Structural relationships for the design of responsive azobenzene-based lyotropic liquid crystals†
Abstract
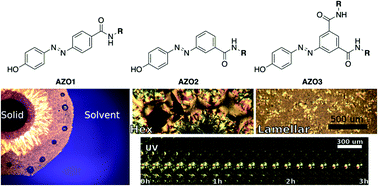
Light-responsive binary (azobenzene + solvent) lyotropic liquid crystals (LCs) were investigated by structural modification of simple azobenzene molecules. Three benzoic acid-containing azobenzene molecules 4-(4-(hydroxyphenyl)diazenyl)benzoic acid (AZO1), 3-(4-(hydroxyphenyl)diazenyl)benzoic acid (AZO2) and 5-(4-(hydroxyphenyl)diazenyl)isophthalic acid (AZO3) were produced with various amide substitutions to produce tectons with a variety of hydrophobicity, size and branching. The LC mesophases formed by binary (azobenzene + solvent) systems with low volatility solvents dimethylsulfoxide (DMSO) and N,N-dimethylformamide (DMF) as well as the protic ionic liquids ethylammonium formate (EAF) and propylammonium formate (PAF), were investigated using a combination of small-angle X-ray and neutron scattering (SAXS and SANS) as well as polarising light microscopy (PLM). Increasing alkyl group length was found to have a strong influence on LC phase spacing, and changes in the position of substitution on the benzene ring influenced the preferred curvature of phases. UV-induced trans to cis isomerization of the samples was shown to influence ordering and optical birefringence, indicating potential applications in optical devices.


 Please wait while we load your content...
Please wait while we load your content...