Ordering self-assembly structures via intermolecular Br⋯S interactions†
Abstract
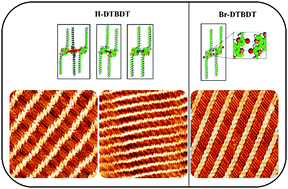
Recent research studies have shown that the halogenated benzo[1,2-b:4,5-b′]dithiophene (DTBDT) unit as a polymer donor exhibits high charge carrier mobility due to the well-ordered molecular packing and high crystallinity, which is meaningful for achieving highly efficient organic solar cells (OSCs). However, it is difficult to acquire the accurate packing information of polymer materials. Herein, we investigated the self-assembled behaviors of two DTBDT derivatives, 4,8-bis(4-octadecylthiophen-2-yl)benzo[1,2-b:4,5-b′]dithiophene (H-DTBDT) and 4,8-bis(5-bromo-4-octadecylthiophen-2-yl)benzo[1,2-b:4,5-b′]dithiophene (Br-DTBDT), to elucidate the effect of introducing a bromine atom on molecular packing structures by STM at the 1-phenyloctane/HOPG interface. It is observed that the H-DTBDT molecules exhibit a random arrangement along each lamella, while the Br-DTBDT molecules self-assemble into a highly ordered lamellar structure. Density functional theory (DFT) analysis combined with the topological properties of the electron density at the bond critical points revealed that the existence of weak intermolecular interactions of Br⋯S facilitates the regular packing motif of Br-DTBDT molecules. The results helped us to understand that the Br⋯S bond generally acted as the auxiliary force and can play the primary role in the construction of supramolecular nanostructures.


 Please wait while we load your content...
Please wait while we load your content...