Selective nucleation of ice crystals depending on the inclination angle of nanostructures†
Abstract
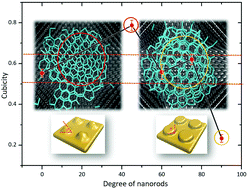
Heterogeneous nucleation is decided by many factors, and surface morphology is one of the most important elements. This paper reports the selective ice nucleation and growth process on a series of nanorods with different inclinations, which were rarely mentioned in previous research studies. It is found that the nanorods with special inclinations can cause the selective nucleation of ice crystals because of the spatial geometry matching. On this basis, we can regulate the ice crystal types (mainly including cubic ice and hexagonal ice) accordingly and even improve the freezing efficiency via controlling the inclinations of surface nanorods. In particular, cubic ice occupies the dominant role in the ice crystal on the surface of 45°-inclination nanorods, yet 90°-inclination nanorods are more beneficial for the formation of hexagonal ice. The shape of the nanorods not only controls the type of ice crystal, but also changes the freezing efficiency because different ice crystals have an unequal nucleation energy barrier. There are no apparent differences in the freezing efficiency on nanostructures with 45°, 75° and 90° inclination nanorods, and 60°-inclination nanorods are more favorable for ice nucleation. Our studies can promote the understanding on the selective nucleation of ice crystals and provide a theoretical basis for achieving the regulation of freezing efficiency.


 Please wait while we load your content...
Please wait while we load your content...