Solvation of cholesterol in different solvents: a molecular dynamics simulation study
Abstract
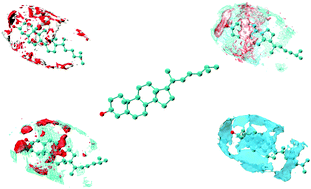
To the best of our knowledge, molecular dynamics simulations of an isolated cholesterol immersed in four different solvents of varying polarity, such as water, methanol, dimethyl sulfoxide and benzene, were reported for the first time to gain insights into the structural and dynamical properties. The study was mainly focused on the evaluation of solvation of cholesterol with respect to its hydrophilic and hydrophobic structural components in the form of respective functional groups interacting with the solvents. Structural evaluations suggested that both hydrophilic and hydrophobic groups of cholesterol were interacting with the solvents, in particular methanol and dimethyl sulfoxide, which presented both types of interactions that are polar and non-polar. On the other hand, the highly polar water and non-polar benzene demonstrated extreme solvation behavior, since water was involved only in hydrogen bonding to the solute hydroxyl group and non-polar benzene formed strong van der Waals interactions only. Furthermore, the hydrophobic effect of cholesterol was also analyzed mainly in polar solvents, as the effect was more pronounced in the polar environment thereby preventing the solvent mobility in the solvation layer(s). The dynamical properties in terms of lateral diffusion and hydrogen bond dynamics as well as free energies of solvation also corroborated the findings based on the structural data and the hydrophobic character of cholesterol was later quantified by the computation of the averaged solvent accessible surface area. The polarity effect of the solvents on the aggregation property of cholesterol was further investigated, which is of big concern from the clinical point of view due to its major role in cardiovascular ailments. It was another major finding of the present study that aggregation was shown to be facilitated by highly polar solvents like water.


 Please wait while we load your content...
Please wait while we load your content...