Re-evaluating the role of phosphinic acid (DINHOP) adsorption at the photoanode surface in the performance of dye-sensitized solar cells
Abstract
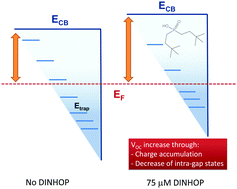
Dineohexyl phosphinic acid (DINHOP) is a popular amphiphilic molecular insulator considered as the most efficient co-adsorbent (co-grafter) for the improvement of the photovoltaic performance of TiO2 based hybrid solar cells. Although the effect of its incorporation on the improvement of cell performance has been well demonstrated, the mechanisms through which it affects the photovoltaic and electrodynamic parameters of the cells are not yet clear. Here we re-examine the mechanism through which the DINHOP co-adsorbent affects the photovoltaic and electrodynamic parameters of dye-sensitized solar cells. Although DINHOP is widely believed to inhibit (passivate) recombination across the TiO2/electrolyte interface, we demonstrate that this is sublte, noticeable only for a very high concentration (e.g. 750 μM) of DINHOP, co-sensitized with a dye. For the most frequently used DINHOP concentrations (e.g. 75 μM and 375 μM), an observed increase of the diffusion coefficient and recombination rate could be directly associated with a decrease of total intra-gap states in TiO2. For a DINHOP concentration as low as 75 μM, the conduction band edge of TiO2 moves upward due to the combined effect of charge accumulation and a decrease in the total number of intra-gap states leading to an effective enhancement of the DCCS VOC, where the decrease in total intra-gap states does not contribute positively. The decrease of total intra-gap states enhances both the transport and recombination rates of charge carriers by the same fraction due to a transport-limited recombination process. On the other hand, adsorption of DINHOP molecules at higher concentrations such as 375 μM and 750 μM additionally modifies the distribution of intra-gap states, affecting the nonlinear recombination parameter of charge carriers at the anode–electrolyte interface, leading to an overall enhancement of the DSSC VOC. In all cases, incorporation of DINHOP results in an overall improvement of the solar cell efficiency (∼14% compared with the reference one), with a maximum for a concentration of 375 μM, where no inhibition of recombination was observed. Interestingly, for this DINHOP concentration, we estimate that 1 DINHOP molecule per every 12 molecules of dye occupies the intra-gap states of the TiO2 surface. The results presented in this work elucidate the physical phenomena involved in the interaction of co-adsorbents, pre-treatments or additives with the electrolyte at the surface of the TiO2 photoanode of dye-sensitized solar cells and can be easily adapted to study other electrochemical systems.


 Please wait while we load your content...
Please wait while we load your content...