Exact quantum dynamics background of dispersion interactions: case study for CH4·Ar in full (12) dimensions†
Abstract
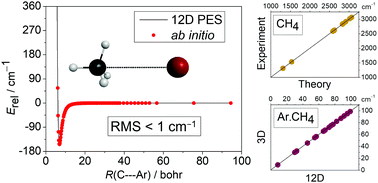
A full-dimensional ab initio potential energy surface of spectroscopic quality is developed for the van-der-Waals complex of a methane molecule and an argon atom. Variational vibrational states are computed on this surface including all twelve (12) vibrational degrees of freedom of the methane–argon complex using the GENIUSH computer program and the Smolyak sparse grid method. The full-dimensional computations make it possible to study the fine details of the interaction and distortion effects and to make a direct assessment of the reduced-dimensionality models often used in the quantum dynamics study of weakly-bound complexes. A 12-dimensional (12D) vibrational computation including only a single harmonic oscillator basis function (9D) to describe the methane fragment (for which we use the ground-state effective structure as the reference structure) has a 0.40 cm−1 root-mean-square error (rms) with respect to the converged 12D bound-state excitation energies, which is less than half of the rms of the 3D model set up with the 〈r〉0 methane structure. Allowing 10 basis functions for the methane fragment in a 12D computation performs much better than the 3D models by reducing the rms of the bound state vibrational energies to 0.07 cm−1. The full-dimensional potential energy surface correctly describes the dissociation of the system, which together with further development of the variational (ro)vibrational methodology opens a route to the study of the role of dispersion forces in the excited methane vibrations and the energy transfer from the intra- to the intermolecular vibrational modes.
- This article is part of the themed collection: 2020 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...