Mechano-chemical stability and water effect on gas selectivity in mixed-metal zeolitic imidazolate frameworks: a systematic investigation from van der Waals corrected density functional theory†
Abstract
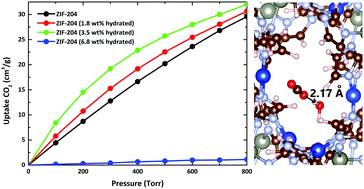
A series of Zn/Cu Zeolitic Imidazolate Frameworks (ZIFs) ZIF-202, -203, and -204 are systematically investigated by Density Functional Theory (DFT) with and without van der Waals (vdW) corrections. The elastic constants for non-solvent structures indicate that ZIF-202 and -204 are mechanically stable while ZIF-203 is unstable, which arises from the stiffness along the x-axis under a uniaxial strain in the PBE-D3 method. By considering the presence of solvents in ZIF-203, a structural phase transformation from a monoclinic to a triclinic structure is found which could be explained by the Jahn–Teller distortion. From the chemical bonding point of view, it is found that vdW interactions and hybridization between d-orbitals (copper) and p-orbitals (imidazolate) are the main-driving forces in stabilizing ZIF-202 and -204, respectively. The electronic structure calculations predict the presence of two optical transitions in the visible region in agreement with the experimental observation for ZIF-204 both without and with water. The DFT simulations reveal that CO2 molecules prefer to locate near imidazolate and water in dry and hydrated ZIF-204, respectively. The analysis of Canonical Monte Carlo (GCMC) simulations reveals that Coulomb interaction between CO2 and H2O molecules is mainly responsible for the enhanced CO2 uptake and selectivity under humid conditions compared to dry ones.


 Please wait while we load your content...
Please wait while we load your content...