Progress on cocrystallization of poorly soluble NME's in the last decade
Abstract
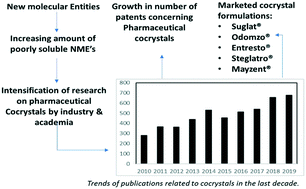
For tailoring the physicochemical properties of active pharmaceutical ingredients, cocrystallization has been an exciting field of interest to the researchers. As in recent years, due to the prevalence of poorly soluble pharmaceuticals that demonstrate poor and erratic bioavailability, pharmaceutical cocrystals' applicability to alter the physicochemical properties has gained attention. This encouraged different regulatory bodies to draft regulatory guidelines for pharmaceutical cocrystals, which were followed by approval for marketed pharmaceutical cocrystals in different countries. In this article, a review of the cocrystals formed by the poorly soluble new molecular entities (NME's) approved by the FDA and the other approved marketed cocrystal formulations for therapeutical use in the last decade (2010–2019) is provided. The cocrystals of the NME's were studied from different patent databases and the publications, and the advantages that the cocrystals possess over the NME's are reported here. This article could also act as an informative record to the readers for the imminent formation of new cocrystals from these NME's by guiding with the formulation techniques and selection of the coformers that have already been reported.
- This article is part of the themed collections: Database Analysis and 2020 Highlight article collection


 Please wait while we load your content...
Please wait while we load your content...