Evaluating the role of a urea-like motif in enhancing the thermal and mechanical strength of supramolecular gels†
Abstract
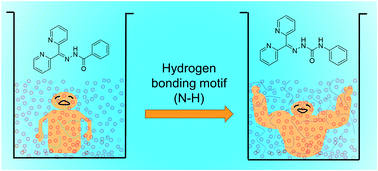
The geometry and spatial orientation of gelator molecules and the mode of various intermolecular non-covalent interactions are the key parameters that dictate the structure and properties of low molecular weight gelators (LMWGs). The effect of intermolecular non-covalent interactions in tuning the gelation properties was analysed for dipyridyl hydrazone (HL1) with an amide-like hydrogen bonding moiety and semicarbazone (HL2) with a urea-like motif. The gelation properties of the hydrazone and semicarbazone compounds were studied in a series of solvents and solvent mixtures and the SEM images of the xerogels revealed that the morphology of the HL2 gelator was more fibrous in nature compared to HL1. The mechanical and thermal stability of HL2 was higher than HL1, which was confirmed by rheology and gel–sol transition temperature experiments, respectively. The key non-covalent interactions responsible for gel formation were assigned using X-ray diffraction techniques and the results were corroborated with the gelation properties. The stimuli-responsive properties of the gelators were studied by analysing the effect of metal salts and anions on the gelation properties of HL1 and HL2 and the results indicated that metal complexation disrupted the gel network whereas the addition of anions did not alter the gelation ability of the gelators. The tuning of gelation properties by metal complexation and the comparison of intermolecular non-covalent interactions of the gelators enabled us to identify the key parameters responsible for gel-network formation in HL1 and HL2.
- This article is part of the themed collection: Supramolecular & Polymorphism


 Please wait while we load your content...
Please wait while we load your content...