Precise control of density and strength of acid sites of MFI-type zeolite nanoparticles via simultaneous isomorphous substitution by Al and Fe†
Abstract
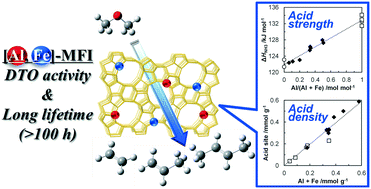
MFI-type zeolite nanoparticles substituted isomorphously by both Al and Fe and of crystallite size ≤51 nm were prepared via a hydrothermal synthesis method combined with a pre-heating treatment. Quantitative assessment of the density and strength of acid sites of the synthesized MFI-type zeolites using temperature-programmed desorption of NH3 demonstrated that the strength and density of the acid sites on the synthesized zeolites were linearly dependent on the contents and relative ratios of Al and Fe, and the acidities of the [Al, Fe]-MFI-type zeolites were between those of [Al]-MFI and [Fe]-MFI. The observed direct correlations also indicated that the density and strength of acid sites of the MFI-type zeolites were tunable by simply altering the relative amounts of reagents used. Such controlled and moderate acidity for the [Al, Fe]-MFI-type nanoparticles facilitated the suppression of coke formation in the catalytic conversion of dimethyl ether to light olefins.


 Please wait while we load your content...
Please wait while we load your content...