Hydrothermal synthesis of BaLu2F8:Ln3+ crystals: phase/morphology evolution, energy transfer and tunable multicolor luminescence†
Abstract
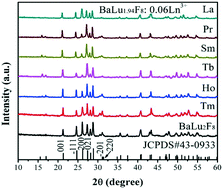
BaLu2−xF8:xLn3+ crystals have been synthesized via a one-step hydrothermal route without any surfactants. X-ray diffraction (XRD), scanning electron microscopy (SEM), and down-shift/up-conversion photoluminescence spectroscopy were used to characterize the samples. Pure monoclinic BaLu2F8 crystals were obtained at pH = 7 with NaBF4 as a fluoride source. Doping 0.06 mmol Ln3+ ions from Ho3+ to La3+ and doping Yb3+ ions from 0.1 to 0.4 mmol both have caused dramatic changes to the product's morphology from prismatic microrods of about 10 μm to particles of about 3 μm aggregated by flakes. The differences in the phase, size and shape of the final products were greatly related to the fluoride source and pH values, which have been studied in detail. In addition, surfactants also affected the phase and morphology of the BaLu2F8 crystals. Multicolor down-shift emission was achieved in the BaLu1.94F8:0.06Ln3+ (Ln = Sm, Eu, Tb, Dy, Er and Tm) crystals under UV lamp excitation at 365 nm, and multicolor UC luminescence has been achieved in BaLu2F8:Yb3+,Ln3+ (Ln = Er, Tm, Ho) systems. When the Yb3+–Er3+ ions are co-doped into the host, the mechanism for the red emission (4F9/2 → 4I15/2) retains the tri-photon process like the single-doped Er3+ ion, but the mechanism for the green emission (2H11/2/4S3/2 → 4I15/2) is a two-photon process. Furthermore, the UC luminescence mechanism for the co-doped Yb3+–Ho3+ pairs is also a tri-photon process in the BaLu2F8 matrix. The as-prepared BaLu2F8:Ln3+ phosphors have potential applications in the fields of light emitting devices, three-dimensional displays and backlights.


 Please wait while we load your content...
Please wait while we load your content...