2-Mercaptoimidazolium halides: structural diversity, stability and spontaneous racemisation†
Abstract
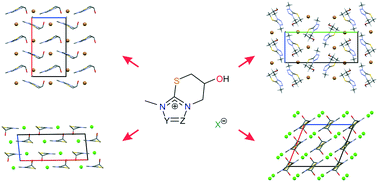
Experimental and theoretical characterisation and studies of the stability of heterobicyclic thiazinium salts (bicyclic 2-mercaptoimidazolium chlorides and bromides) were performed to rationalise and understand the influence of the counterion (Cl− ↔ Br−) and the replacement of CH by N on crystal packing, the influence of the anion on the moisture and temperature dependent stability, and the racemisation behaviour of the imidazo-thiazinium chloride. Six compounds were synthesised and for five of the compounds the structures were solved from single-crystal X-ray diffraction data. The structural features of the sixth compound could be derived from powder X-ray diffraction data comparisons. An exchange of the Cl− anion by Br− does not influence the crystal packing of the racemic thiazinium salt but increases its moisture dependent stability. In contrast, replacing the imidazole moiety of the cation by a triazole or tetrazole moiety results in distinct packing arrangements of the investigated bromide salts, although, substitution calculations suggest that isostructural packing arrangements might exist. The binary melting point phase diagram was constructed to confirm the nature of the racemic species of the thiazinium chloride, and differential scanning calorimetry and lattice energy minimisations were used to estimate the enthalpy difference between the racemic and enantiopure crystals, rationalising the high tendency of racemisation of the enantiopure compound.


 Please wait while we load your content...
Please wait while we load your content...