A carbon network strategy to synthesize silicon–carbon anodes toward regulated morphologies during molten salt reduction†
Abstract
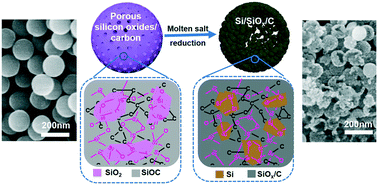
Low temperature molten salt reduction has been demonstrated as the most intriguing approach to prepare silicon–carbon hybrid anode materials due to its cost-effectivity and scalability. However, a common issue plaguing this method is the controllability of product morphology. Herein, a carbon network strategy was proposed to explore the influence of carbon distribution in silicon oxide/carbon nanocomposites on the morphology transition during molten salt reduction. The result shows that the densification of the three-dimensional carbon framework affects the kinetics of the reduction process, thus achieving distinct morphologies of the final product. An appropriate carbon network is conducive to forming porous Si/SiOx/C hollow spheres, while a dense carbon network will hinder the internal diffusion and form a pitaya-like structure. When the carbon network is too sparse or without carbon, the reduction products tend to collapse to form irregular aggregates. As a lithium-ion battery anode, the Si/SiOx/C nanocomposite with a hollow sphere structure manifests outstanding electrochemical performance in terms of superior cycling stability and excellent rate performance. The current work can provide new insights into the preparation of carbon–silicon based composites with various morphologies via the molten salt method.


 Please wait while we load your content...
Please wait while we load your content...