One-dimensional arrangement of NORIA in the solid-state†
Abstract
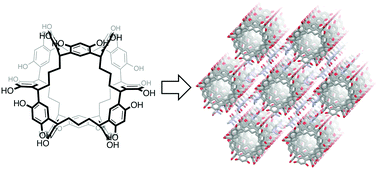
NORIA is a synthetic macrocycle consisting of twelve resorcinol rings. The hydroxy groups surrounding the surface of NORIA make it difficult for NORIA to be dissolved in organic solvents, except for polar solvents, a property which is ideal for developing solid-state materials. For developing solid-state materials, information about the effect of the crystallization conditions on the crystal packing of NORIA is required, although only limited examples have been reported so far. We find that the crystal packing of NORIA depends heavily on the crystallization conditions. Single crystals are grown in DMF solutions by the slow diffusion of pentane, cyclohexane, methylcyclohexane, benzene, and toluene into the solution. X-ray diffraction analysis demonstrates that the saturated hydrocarbon vapors induce the molecules to organize into a triclinic crystal packing, whereas another crystal packing, wherein NORIA is arranged linearly in the hexagonal crystal, is formed in the case of aromatic vapors. In the five crystals, DMF molecules are trapped by NORIA and interact with neighboring hosts through hydrogen bonding, indicating that the DMF molecules function as linkers. The X-ray crystal structures suggest that the planarity of the guests is a factor behind the formation of the hexagonal crystal packing. Electron density maps and the solvent accessible surface of NORIA suggest that small molecules, such as water, can access the cavity. The large solvent-accessible volumes in the unit cells of both crystal packings demonstrate that NORIA functions as an excellent building block for lattice inclusion compounds.


 Please wait while we load your content...
Please wait while we load your content...