Engineering CuOx–ZrO2–CeO2 nanocatalysts with abundant surface Cu species and oxygen vacancies toward high catalytic performance in CO oxidation and 4-nitrophenol reduction†
Abstract
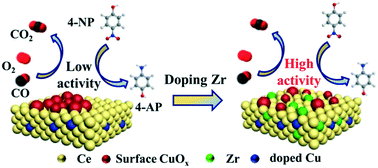
Highly dispersed surface Cu species and oxygen vacancies are important for CuOx–CeO2 catalysts in many reactions, but it is difficult to increase the concentrations of highly dispersed Cu species and oxygen vacancies simultaneously by only adding a Cu component. Herein, CuOx–ZrO2–CeO2 (CZC) nanocatalysts with different ratios of Cu/Zr have been effectively produced by a simple solvent-free synthetic strategy. The effect of Zr doping on the structure and reactivity of the CuOx–CeO2 catalysts has been investigated with the help of carbon monoxide (CO) oxidation and 4-nitrophenol (4-NP) reduction as model reactions. It is revealed that the introduction of Zr relieved the agglomeration of surface Cu species, produced more oxygen vacancies in the interior and Cu+ species on the surface of the catalyst, and increased the reducibility of CuOx–CeO2. The CZC catalysts show remarkably enhanced catalytic activity due to the abundant surface active sites and the improved redox properties provided by the suitable doping of Zr into CuOx–CeO2. This work not only offers a practicable way to reconstruct the distribution of active metals on the metal oxide support but also provides a high-efficiency strategy to prepare oxide catalysts with excellent catalytic performance.
- This article is part of the themed collection: Editor’s Collection: Imperfect nanocrystals for Perfect Catalysis


 Please wait while we load your content...
Please wait while we load your content...