Coordination polymers of paramagnetic bis(leucinato)copper(ii) diastereomers: experimental and computational study of the stereoisomerism and conformations†
Abstract
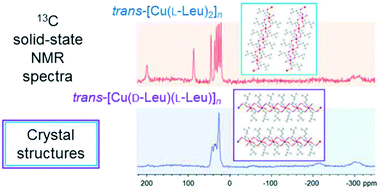
This paper demonstrates the utility of joint applications of X-ray diffraction, solid state (ss) NMR, density functional theory (DFT) and molecular mechanics (MM) calculations for the determination of the stereochemistry of paramagnetic bis(aminoacidato)copper(II) compounds. Solution-based and mechanochemical syntheses of trans-bis(leucinato)copper(II) [Cu(Leu)2] with L- and DL-leucine using different solvents always resulted in the same two-dimensional coordination polymers of the LL (previously reported in the literature) and DL diastereomers. The X-ray crystal and molecular structure of trans-catena-[(μ-D-leucinato)(μ-L-leucinato)copper(II)] (space group C2) has symmetry unrelated D- and L-leucine side-chain conformations. An asymmetrical copper(II) DL-amino acid complex has not been observed so far in a non-centrosymmetric space group. Both LL and DLtrans-[Cu(Leu)2] crystals have similar intermolecular bonding. The X-ray powder diffraction and magic-angle spinning (MAS) ssNMR information available from the polycrystalline bulk phase of the two compounds agree with their single-crystal results. Besides, the 2H MAS ssNMR spectrum of the polycrystalline sample synthesized using DL-leucine showed weak signals which were ascribed to a small amount of the LL (DD) stereoisomers. DFT calculations of the Fermi-contact contributions aided the 13C and 2H NMR spectral assignments. DFT conformational analyses of the LL and DL isomers revealed two Leu side-chain conformations as the energetically favorable ones in the gas phase and aqueous solution. These are exactly the conformations whose combinations occur in the observed crystal structures. Similar MM potential energies of LL and DLtrans-[Cu(Leu)2] in the crystal lattices suggest that crystallization of DL over LL (DD) diastereomers should be attributed to kinetic rather than thermodynamic effects.


 Please wait while we load your content...
Please wait while we load your content...