Photophysical properties of Ag, Zn and Cd - N-(4-pyridylmethyl)-1,8-naphthalimide complexes: influences of π-stacking and C–H⋯O interactions†
Abstract
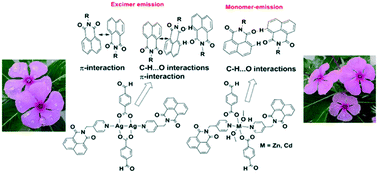
The influence of π-stacking and C–H⋯O interactions on photoluminescence of a dinuclear silver(I), a mononuclear zinc(II) and a cadmium(II) formylbenzoate complex possessing N-(4-pyridylmethyl)-1,8-naphthalimide are reported. The packing patterns of all the complexes have covalently linked naphthalimide dimers held by C–H⋯O interactions, where the participating naphthalimide rings are on a single plane. The silver complex has an additional naphthalimide ring located parallel over each dimer, and the parallel rings are in a head-to-tail π-stacking arrangement, and such π-stacking is found in the ligand also. Meanwhile, parallel cofacial arrangements among aromatic rings of naphthalimide and formylbenzoate are observed in zinc and cadmium complexes. Silver complex has emission at 515 nm, comparable to the emission of the free ligand, but zinc and cadmium complexes emit at shorter wavelengths as compared to the ligand. Photophysical properties of the ligand and the complexes in solution are distinguishable from that observed in solid state.


 Please wait while we load your content...
Please wait while we load your content...