Structural variety of aluminium and gallium coordination polymers based on bis-pyridylethylene: from molecular complexes to ionic networks†
Abstract
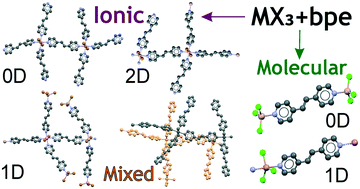
A systematic structural study of the complexes formed by aluminium and gallium trihalides with 1,2-bis(4-pyridyl)ethylene (bpe) was performed. Quantum chemical computations revealed that the energy differences between the ionic and the molecular complexes expected in the MX3–bpe (M = Al, Ga; X = Cl, Br) system are very small. These computational findings indicate that this system is a rich source of compounds with diverse structural motifs. Indeed, eleven complexes, namely, [M2Cl4(bpe)5]2+[M2Cl4(bpe)6]2+[MCl4]−4*3bpe (M![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Al (1), M
Al (1), M![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Ga (2)), [Al3Br8(bpe)3]+[AlBr4]− (3), [Al2Br4(bpe)5]2+[AlBr4]−2*bpe (4), [Ga2Br4(bpe)7]2+[GaBr4]−2*bpe (5), [GaCl3(bpe)]∞ (6), [(MX3)2(bpe)] (MX3 = AlCl3 (7), AlBr3 (8), GaCl3 (9), GaBr3 (10a, 10b), and [(GaBr3)2(bpe)]*bpe (11) were synthesized in a solvent-free melt reaction of group 13 metal halides and bpe. The diversity of the complexes obtained shows the marked effect of different reagent ratios as well as Lewis acid on the product structures. Ionic coordination polymers (CPs) 1 and 2 exhibit isostructural unusual mixed one-dimensional–two-dimensional (1D–2D) networks. Interaction of bpe with excess AlBr3 yielded ionic 1D CP 3 while the same reaction in excess bpe produced ionic 2D CP 4. Reaction of GaBr3 and bpe in equimolar ratio yielded binuclear ionic complex 5 in a mixture with an adduct solvate 11. The only molecular CP 6 with 1D structure was isolated when the reaction between GaCl3 and bpe was carried out in both 1 : 1 and 1 : 2 stoichiometric ratios. In the case of the 2 : 1 ratio, irrespective of the Lewis acid, [(MX3)2(bpe)] adducts 7–10a and 10b were obtained. Remarkably, complex 10 crystallizes in two polymorphic modifications, 10a and 10b. The solid-state structures of complexes 1, 3–5, and 7–11 were determined for the first time.
Ga (2)), [Al3Br8(bpe)3]+[AlBr4]− (3), [Al2Br4(bpe)5]2+[AlBr4]−2*bpe (4), [Ga2Br4(bpe)7]2+[GaBr4]−2*bpe (5), [GaCl3(bpe)]∞ (6), [(MX3)2(bpe)] (MX3 = AlCl3 (7), AlBr3 (8), GaCl3 (9), GaBr3 (10a, 10b), and [(GaBr3)2(bpe)]*bpe (11) were synthesized in a solvent-free melt reaction of group 13 metal halides and bpe. The diversity of the complexes obtained shows the marked effect of different reagent ratios as well as Lewis acid on the product structures. Ionic coordination polymers (CPs) 1 and 2 exhibit isostructural unusual mixed one-dimensional–two-dimensional (1D–2D) networks. Interaction of bpe with excess AlBr3 yielded ionic 1D CP 3 while the same reaction in excess bpe produced ionic 2D CP 4. Reaction of GaBr3 and bpe in equimolar ratio yielded binuclear ionic complex 5 in a mixture with an adduct solvate 11. The only molecular CP 6 with 1D structure was isolated when the reaction between GaCl3 and bpe was carried out in both 1 : 1 and 1 : 2 stoichiometric ratios. In the case of the 2 : 1 ratio, irrespective of the Lewis acid, [(MX3)2(bpe)] adducts 7–10a and 10b were obtained. Remarkably, complex 10 crystallizes in two polymorphic modifications, 10a and 10b. The solid-state structures of complexes 1, 3–5, and 7–11 were determined for the first time.


 Please wait while we load your content...
Please wait while we load your content...