Controlled protein crystal nucleation in microreactors: the effect of the droplet volume versus high supersaturation ratios†
Abstract
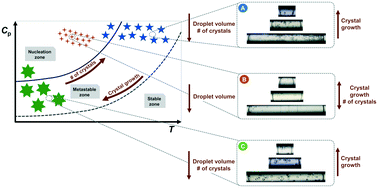
Herein, the control of enhanced protein nucleation in microdroplets under high supersaturation ratios is discussed, where lysozyme is used as a model protein. This study is conducted to evaluate the influence of droplet volume on the size and number of crystals through isothermal and double-pulse temperature experiments performed in a droplet-based microreactor. Lysozyme concentration is estimated during and at the end of the crystallization experiments, based on analytical and experimental results, respectively. Furthermore, nucleation rate in microdroplets is calculated following a Poisson distribution, and crystal growth is assumed to be diffusion-controlled. Controlled and enhanced lysozyme nucleation is achieved in smaller droplet volumes and under certain double-pulse temperature conditions. The droplet volume effect becomes more important at lower supersaturation ratios as the crystal number follows the droplet volume increase, while at higher supersaturation ratios, the crystal number does not considerably vary with the droplet volume. Finally, the proposed analysis can be adopted as an experimental design tool and extended to the crystallization of other macromolecules.


 Please wait while we load your content...
Please wait while we load your content...