Strong stacking interactions at large horizontal displacements of tropylium and cyclooctatetraenide ligands of transition metal complexes: crystallographic and DFT study†
Abstract
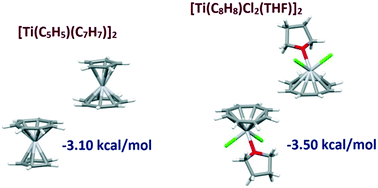
A Cambridge Structural Database (CSD) search was performed in order to study the stacking interactions of the 7-membered tropylium ring and 8-membered cyclooctatetraenide (COT) ring, coordinated to transition metals via their π-electrons. The search showed that both ligands have very high preference for stacking interactions with large horizontal displacements. These interactions leave tropylium and COT faces available for additional simultaneous (mostly C–H/π and in some cases stacking) interactions, which lead to very stable supramolecular structures. DFT calculations on model molecules derived from CSD crystal structures showed that large offset stacking of both tropylium and COT ligands surpasses −3.0 kcal mol−1, which makes them important contributors to the overall stability of the systems they are found in. Small offset stacking of tropylium and COT ligands, which is the strongest type of stacking, is almost not found in the CSD crystal structures, which can be explained based on the fact that they do not enable additional simultaneous interactions with molecules from the environment. Stacking of tropylium and COT ligands occurs at larger offsets than stacking of benzene and cyclopentadienyl ligands, which can be rationalized with their larger size, as well as with their electrostatic potential surfaces.
- This article is part of the themed collection: Database Analysis


 Please wait while we load your content...
Please wait while we load your content...