Dynamic characterization of crystalline fluorophores with conformationally flexible tetrahydrocarbazole frameworks†
Abstract
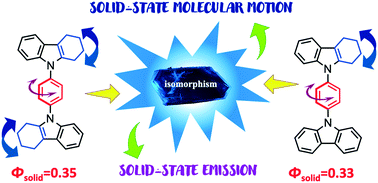
Two new compounds 2 and 3 and their deuterated analogues (2-d4 and 3-d4) were prepared from Ullmann-type coupling reactions. Both molecules show good emissive properties in solution ΦF = 0.13 (2) and 0.57 (3) as well as in the solid-state (ΦF = 0.35 and 0.33, respectively). Variable temperature solid-state NMR measurements using 13C CPMAS and 2H spin-echo helped to identify segmental motion in the phenylene as well as in the aliphatic moieties, which agreed well with the crystallographic disorder in these moieties. The rotational energy barriers for the new compounds were computed with a periodic DFT approach, finding asymmetric rotational potentials with barriers of 3.5 kcal mol−1 (2) and 4.5 kcal mol−1 (3) for 60° jumps and much higher barriers (>25 kcal mol−1) for 180° jumps. Our studies indicate that low frequency 4-fold motions coexist with fluorescence in these crystalline conjugated compounds.


 Please wait while we load your content...
Please wait while we load your content...