A crystalline solid adduct of sulfathiazole–amantadine: the first dual-drug molecular salt containing both antiviral and antibacterial ingredients†
Abstract
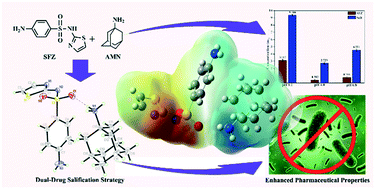
The first molecular salt concurrently combining an antibacterial drug, sulfathiazole (SFZ), and an antiviral constituent, amantadine (ATD), was designed and successfully synthesized via a salification strategy. The precise structure of the newly obtained dual-drug molecular salt SFZ-ATD has been perfectly solved by single-crystal X-ray diffraction and other techniques. The single-crystal diffraction analysis confirmed that the occurrence of proton transfer from SFZ to ATD gave rise to the composition of a molecular salt with an equimolar ratio of SFZ and ATD, where the supramolecular network in the crystal was mainly dominated by charge-assisted one-dimensional hydrogen-bonding chains and a two-dimensional hydrophobic layer. The relevant properties of the molecular salt were subjected to theoretical and experimental studies. The experimental results highlighted that both the permeability and dissolubility of SFZ in the molecular salt were enhanced compared with those of pure SFZ, which was further supported by the theoretical calculations of the molecular electrostatic potential. More importantly, the advantages of the ameliorated physicochemical profile of the dual-drug salt led to intensified antibacterial activities of SFZ against the tested bacterial strains. Thus, this work not only provides a regeneration opportunity for classical sulfa drugs, but also offers new ideas for therapeutic hybridization to treat viruses and bacteria using multidrug combinations obtained via a salification strategy.


 Please wait while we load your content...
Please wait while we load your content...