Kryptoracemic compound hunting and frequency in the Cambridge Structural Database†
Abstract
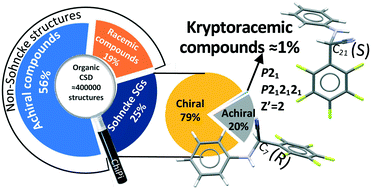
Kryptoracemic compounds (KRCs) are a rare case of racemic pairs of antipodes crystallizing in Sohncke (chiral) space groups. In order to identify KRCs in the Cambridge Crystallographic Structural Database (CSD), a Python script named ChiPi was written to automatically assign the chirality of each crystal structure. The ChiPi code is able to compare each residue contained in a crystal structure based on the chiral centres that were identified and allows discrimination between enantiomeric, diastereomeric, racemic, meso and scalemic structures. It was used to process 393 012 organic entries from the CSD corresponding to almost the entire set of organic crystal structures. It is estimated that racemic compounds constitute 23.8% and 22.2% of centrosymmetric and achiral non-centrosymmetric organic structures in the CSD, respectively. The KRCs represent 0.2% of the whole database and 0.8% of the chiral space groups. The KRC occurrence represents circa 1% (724 structures) of the set of racemic compounds. The distribution of the KRC space groups is drastically shifted toward lower symmetry space groups with a large prevalence of P21 structures. This trend is not restricted to KRCs only but can be extended to structures containing chiral molecules with an even Z′ number.
- This article is part of the themed collections: Database Analysis and The Cambridge Structural Database - A wealth of knowledge gained from a million structures


 Please wait while we load your content...
Please wait while we load your content...