Peculiarities of Br⋯Br bonding in crystal structures of polybromides and bromine solvates†
Abstract
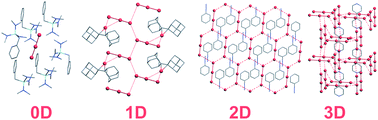
The crystal structures of 204 polybromides and bromine solvates were extracted from the Cambridge Structural Database and the Inorganic Crystal Structures Database. The electron density distribution was theoretically obtained for 18 selected compounds and topological analysis of the electron density by means of the R. Bader atoms in molecules (AIM) theory was performed. For the 18 compounds, the topology of networks formed by Br–Br and Br⋯Br interactions was revealed only within the AIM approach and compared with that found by means of the Voronoi tessellation. Satisfactory correlation between the description of Br⋯Br bonding within the AIM and the Voronoi approaches was found. The effect of cation size, presence of C–Br bonds and bromine content on the dimensionality of the network was studied for 204 solids using the Voronoi tessellation. The Voronoi approach demonstrated good potential to reveal extended architectures based on Br⋯Br interactions that are typical for electroconductive polybromide-based materials.
- This article is part of the themed collections: Database Analysis and The Cambridge Structural Database - A wealth of knowledge gained from a million structures


 Please wait while we load your content...
Please wait while we load your content...