Diminishing accessibility of electrophilic nickel(ii) centres due to incorporation of a methylene spacer in the pendant side arm of a series of heterotrinuclear nickel(ii)/sodium complexes: a DFT study using a homodesmotic equation†
Abstract
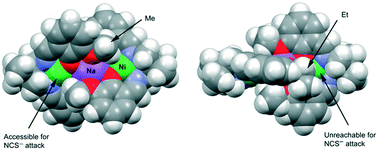
Two compartmental N2O4 donor Schiff bases, H2L1 [N,N-bis(3-methoxysalicylidene)-2,2-dimethylpropane-1,3-diamine] and H2L2 [N,N-bis(3-ethoxysalicylidene)-2,2-dimethylpropane-1,3-diamine], each having inner N2O2 and outer O4 compartments, have been used to prepare four heterotrinuclear nickel(II) complexes [Ni(L1)Na(L1)Ni(NCS)]·H2O (1), [Ni(L1)Na(L1)Ni(N3)] (2), [Ni(L2)Na(L2)Ni]NCS·CH3OH (3), and [Ni(L2)Na(L2)Ni]ClO4 (4). All the complexes have been characterized by elemental and spectral analyses. Single crystal X-ray diffraction analyses have confirmed their structures. In each complex, nickel(II) is placed in the inner N2O2 compartment and sodium is placed in the outer O4 compartment of the respective Schiff base ligand. In complexes 1 and 2, one nickel(II) centre is tetra-coordinated (square planar) and the other is penta coordinated (square pyramidal), whereas in complexes 3 and 4, both nickel(II) centres are tetra-coordinated. The geometries of complexes 1 and 3 have been optimized without a counterion (denoted as 1+ and 3+). The electrophilic nickel(II) centre is found to be accessible in complex 1+ and, conversely, it is unreachable in complex 3+, in agreement with the experimental result. Starting from the hypothetical 1+ complex where both nickel(II) centres are tetra-coordinated, the energy change for replacing four methyl groups with four ethyl groups has been computed using ethane and methane to complete the homodesmotic equation. The result is that the intra-molecular interaction of the four ethyl groups favours complex 3+ by ΔE = −26.1 kcal mol−1.


 Please wait while we load your content...
Please wait while we load your content...