Charge assisted halogen and pnictogen bonds: insights from the Cambridge Structural Database and DFT calculations†
Abstract
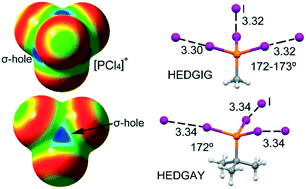
This manuscript combines a search in the Cambridge Structural Database (CSD) and theoretical density functional theory (DFT) calculations to analyse the existence and importance of charge assisted pnictogen and halogen bonds involving halophosphonium cations. Trivalent pnictogen atoms typically have three σ-holes and are able to establish up to three pnictogen bonds (PnBs). In phosphonium salts, the phosphorus atom forms four covalent bonds and, consequently, four σ-holes are located at the extension of these bonds. Therefore, up to four charge assisted PnBs can be formed between these holes and the counterions or any electron rich atom. The covalent bond arrangement around the phosphorus atom is similar to that around tetravalent tetrel atoms and converts into a similar pattern of σ-hole interactions. We have found and described this type of charge assisted pnictogen bond in various halophosphonium crystal structures. Moreover, the competition of charge assisted PnBs with charge assisted halogen bonds (HaBs) has been also studied both theoretically and by analysing the CSD.
- This article is part of the themed collection: The Cambridge Structural Database - A wealth of knowledge gained from a million structures


 Please wait while we load your content...
Please wait while we load your content...