Two-step route to size and shape controlled gibbsite nanoplates and the crystal growth mechanism†
Abstract
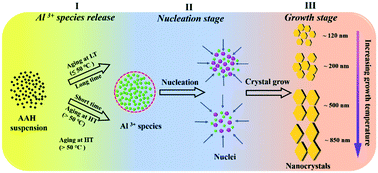
Mastery of gibbsite (α-Al(OH)3) nucleation and crystal growth is significant in fundamental aluminum chemistry and also beneficial for industrial applications and the remediation of nuclear waste. Herein, we developed an additive-free two-step route to size and morphology controlled synthesis of gibbsite nanoplates. Independent temperature manipulation of nucleation and crystal growth regimes was used to control the efficacy and yield of the process, producing gibbsite nanoplates with tunable morphologies and sizes. We achieve morphologies ranging from hexagon, pseudo hexagon, truncated triangle to rhombic, and average basal plane diameters from 120 to 850 nm, with thicknesses between 8 and 45 nm. Analysis of gibbsite structure, size and morphology with techniques including powder X-ray diffraction (p-XRD), 27Al magic angle spinning nuclear magnetic resonance spectroscopy (MAS NMR), scanning electron microscopy (SEM), and atomic force microscopy (AFM) provided key details on the nucleation and crystal growth mechanisms. The results indicate the temperature of the nucleation aging step is the most important factor controlling the final particle size. Particularly small nanoplates were obtained with a low nucleation aging temperature followed by high crystal growth temperature, suggesting that gibbsite growth can be approximated via a LaMer-type burst nucleation and crystal growth mechanism. Scalability was demonstrated by triplicate of 2 L experiments which prepared gibbsite nanoplates of ∼200 nm size and with an average yield of ∼55% after 84 h. Compared to existing techniques, this improved two-step route is cheaper, more efficient and environmentally friendly.


 Please wait while we load your content...
Please wait while we load your content...