Interrelation of thermodynamic sublimation characteristics with crystal structure: adamantane and memantine derivatives of sulfonamide molecular crystals†
Abstract
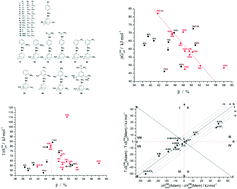
A number of sulfonamide compounds with adamantane (tricyclo[3.3.1.13,7]decane) and memantine (3,5-dimethyladamantan-1-amine) fragments have been synthesized and characterized. Temperature dependencies of saturated vapor pressure of the new molecular crystals were measured by a precise and reproducible inert gas flow method. Based on these results, the standard enthalpies and Gibbs free energies of sublimation at 298.15 K were calculated. The effect of replacement of the adamantane fragment with a memantine one and the change of the substituent in different positions of the phenyl ring for the studied molecules on the thermodynamic characteristics of sublimation was investigated by the diagram method. Correlations between the thermodynamic characteristics of sublimation, melting points and some structural descriptors such as packing density of crystals and free molecular volume were proposed.


 Please wait while we load your content...
Please wait while we load your content...